Microalgae as a source of antimicrobial compounds: A review of bioactive metabolites and their therapeutic awareness
Authors: Arumuganainar Suresh, Email: suresh.a@sssuhe.ac.in
- Apr 10, 2026
- 113 views
Abstract
Microalgae, a varied collection of photosynthetic microorganisms, have become a promising source of bioactive compounds known for their antimicrobial properties. These organisms can transform inorganic carbon-di-oxide (CO2) into biomass while producing an extensive range of primary and secondary metabolites, such as proteins, polysaccharides, lipids, pigments, and polyphenols, that demonstrate antibacterial, antiviral, antifungal, and antiprotozoal effects. As antibiotic-resistant microbes are on the rise, there is an urgent need to explore new treatment options. Microalgae, which have been largely overlooked, could be a promising source of novel antimicrobial compounds. Here, we review microalgae-derived substances that fight off various pathogens, including Gram-positive and Gram- negative bacteria, fungi, viruses, and protozoa. Early findings are promising, but more research is needed to fully understand these compounds, improve their production, and confirm their safety and efficacy in real-world medical use. The review highlights the potential of microalgae as a key tool in fighting infections and calls for continued research into their bioactive properties.
Full Text
Graphical Abstract
Introduction
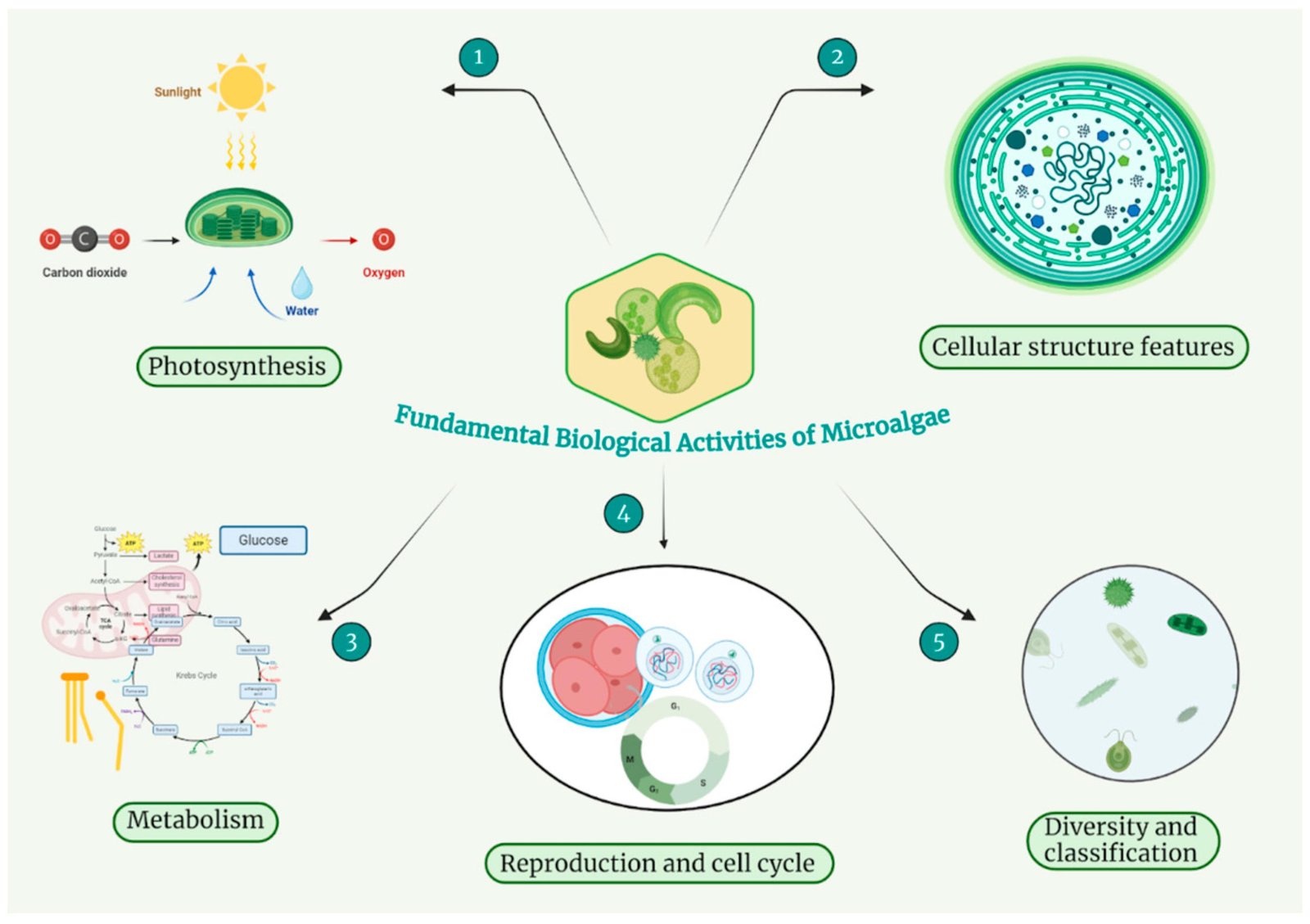
Microalgae, a varied collection of microscopic tiny (1-100µm) phytoplankton, are photosynthetic entities that can transform inorganic carbon dioxide into biomass utilizing water and light energy, thereby playing an essential role in the global carbon cycle1 . Microalgae play a crucial role in ecosystems and are a goldmine of bioactive compounds with huge medical and pharmaceutical potential2. Their biomass is packed with proteins, polysaccharides, lipids, healthy fats, fibers, pigments, vitamins, and minerals making them useful across many industries3 . They also produce powerful secondary metabolites like sterols, lectins, polyphenols, terpenes, and peptides, which have wide-ranging biological effects4 . Compounds from microalgae show real promise in fighting bacteria, viruses, fungi, and parasites5,6. For instance, some microalgae-derived molecules have proven effective against antibiotic-resistant bacteria7 , viruses, fungi8 , and even protozoan infections9 , making them exciting candidates for new treatments
With antibiotic resistance on the rise and few new drugs in the pipeline, the hunt for alternative antimicrobials has put microalgae in the spotlight. These tiny organisms are among Earth’s oldest life forms, and humans have used them for millennia. Historical records show the Chinese relied on microalgae as a famine food over 2000 years ago, while the Aztecs included them in their diet, as noted by Spanish chroniclers10,11. Today,
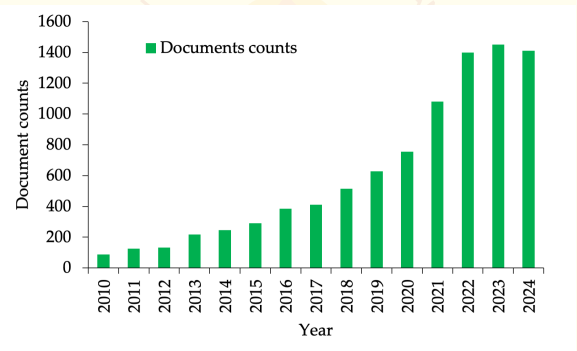
we know microalgae are incredibly diverse with roughly ~1 million species, including both cyanobacteria and eukaryotic varieties. More than 50,000 species have been identified to date in oceans, lakes, and rivers12. Despite this diversity and their advantages over other microbes, the potential of microalgae as a source of bioactive compounds remains largely untapped. In the last decade, there has been a significant surge in interest, with a 3.5-fold increase in research publications focusing on the antimicrobial properties of microalgae (Figure 1).
This review offers an overview of the germ-fighting abilities of microalgae. It brings together recent breakthroughs in their use against hard-to-treat bugs, including ESKAPE bacteria and new fungal threats like mucormycosis. Unlike other reviews which focused on specific types of compounds or single pathogens, our work covers a wide range of microalgal products (peptides, fatty acids, polysaccharides, phenolics, and pigments) and the way they work against bacteria, fungi, viruses, and protozoa. This review also tackles cutting-edge topics such as gene tweaking and improving production processes giving a forward-looking view on how to turn microalgal research into real-world medical and farming uses. This big-picture approach highlights the untapped potential of microalgae as a lasting source of new germ-fighters in the face of worldwide antibiotic resistance.
Figure 1. Number of documents counts on "Microalgae" and "Antimicrobials" for the period of 2010-2025 from the Google scholar database using custom range, accessed 12th March 2025 (https://scholar.google.com/).
2. Microalgal Antimicrobial Compounds and Mode of Action
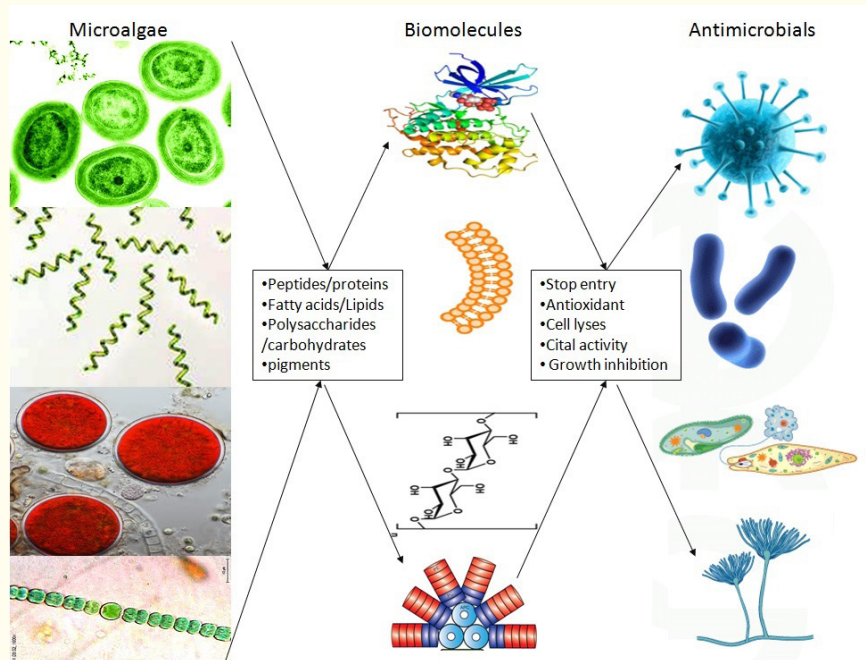
The rising incidence of antibiotic-resistant pathogens has prompted the exploration of new antimicrobial agents, with microalgae emerging as a largely underutilized source of such compounds. Their capacity to flourish in a variety of often harsh environments has led to the development of distinctive biochemical pathways, which in turn produce defensive compounds (both primary and secondary metabolites) that exhibit significant antimicrobial properties13,14,15. These metabolites, including peptides, fatty acids, polysaccharides, phenolic compounds, and pigments, have different modes of action (Table 1 and Figure 2), and demonstrate efficacy against a wide range of pathogens, encompassing bacteria, fungi, parasites, and viruses15,16.
Table 1. Antimicrobial compounds produced by microalgae and their modes of action
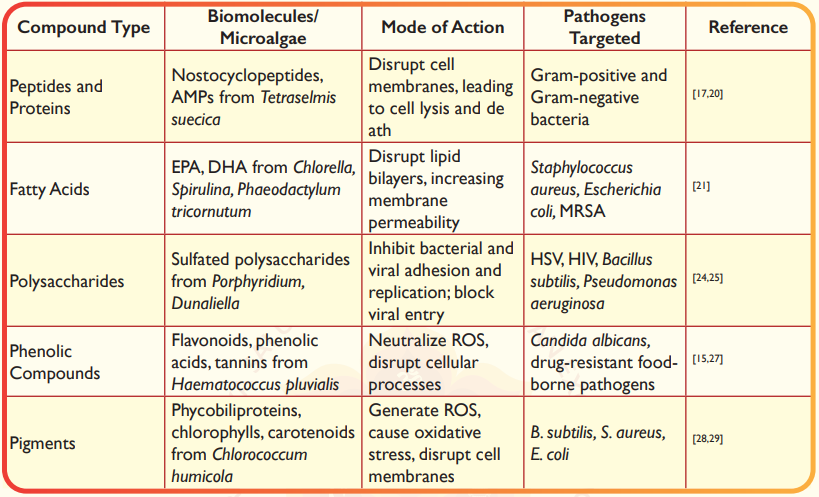
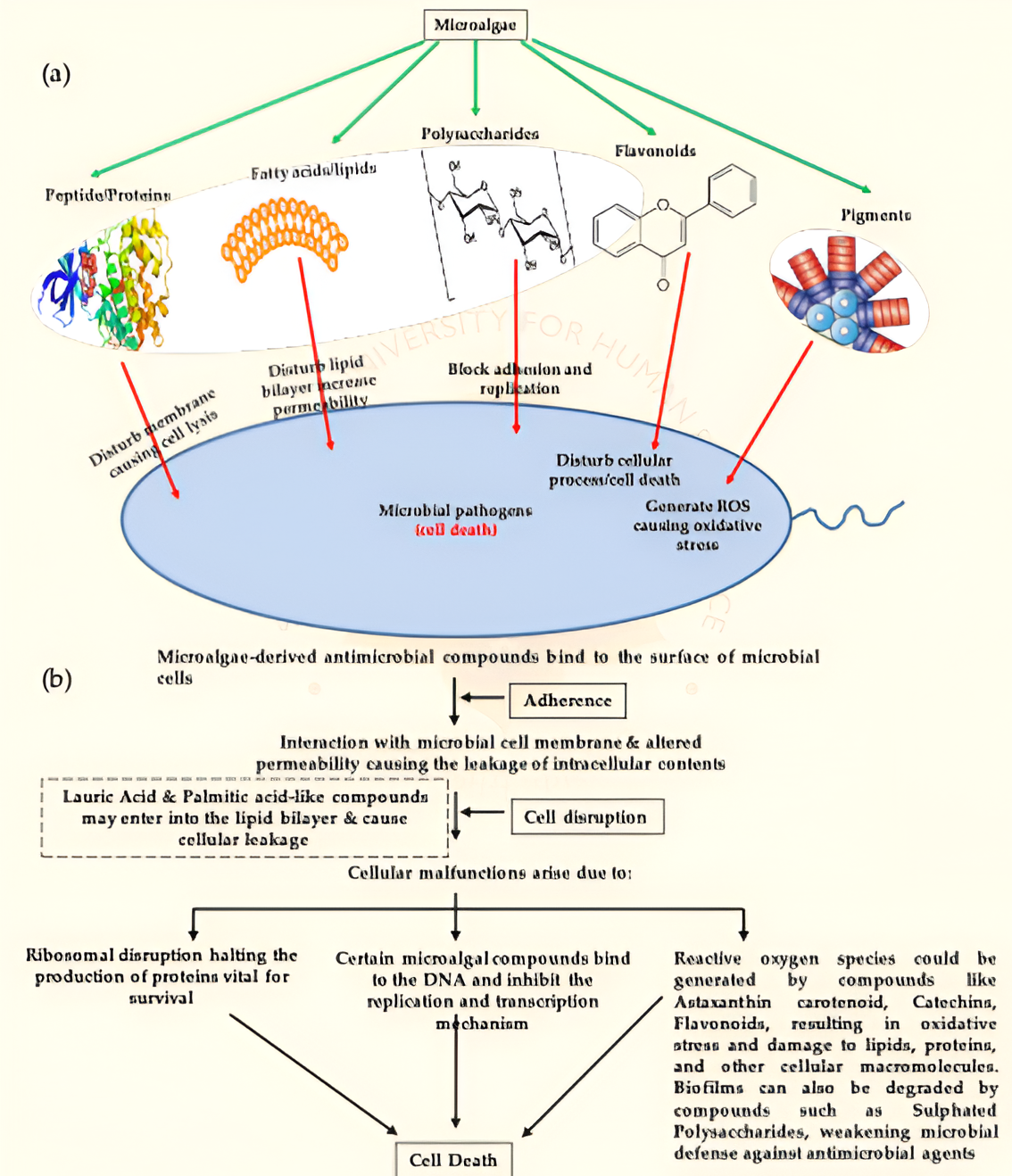
Figure 2. Microalgal antimicrobial compounds (a, b) and their mode of action
2.1 Peptides and Proteins
Microalgae are recognized for their ability to synthesize antimicrobial peptides (AMPs), which are small, positively charged molecules essential to the innate immune defense of these organisms. AMPs generally exert their antimicrobial properties by disrupting the cell membranes of pathogens, resulting in cell lysis and subsequent death17,18. For instance, the cyanobacterium Nostoc produces nostocyclopeptides, a category of cyclic peptides that have shown significant antibacterial efficacy against various Gram-positive and Gram-negative bacteria. Additionally, research by Guzman et al.20 revealed that AMPs extracted from the microalga Tetraselmis suecica also demonstrated antibacterial properties.
2.2 Fatty Acids
Polyunsaturated fatty acids (PUFAs) derived from microalgae, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), have demonstrated notable antimicrobial effects. These fatty acids compromise the integrity of microbial cell membranes by disrupting lipid bilayers, which results in increased permeability and subsequent cell death. For example, microalgae such as Chlorella and Spirulina are recognized for their substantial production of PUFAs, which have been effective in inhibiting the proliferation of various bacterial pathogens, including Staphylococcus aureus and Escherichia coli21. Additionally, EPA and DHA sourced from Phaeodactylum tricornutum have exhibited antimicrobial properties against methicillin-resistant strains of Grampositive S. aureus22. Furthermore, fatty acid extracts from Coccomyxa onubensis have shown inhibitory effects against a diverse array of Gram-positive and Gram- negative bacteria as well as fungi, with the lowest minimum inhibitory concentration (MIC) recorded at 305 and 106 µg/mL against E. coli and Proteus mirabilis, respectively23. The significance of the antimicrobial properties of these fatty acids is underscored by their dual functionality as both nutritional and therapeutic agents.
2.3 Polysaccharides
Sulfated polysaccharides extracted from microalgae, particularly those produced by Porphyridium and Dunaliella, have attracted significant interest due to their extensive antimicrobial properties. These polysaccharides impede the proliferation of bacteria and viruses by disrupting their adhesion and replication mechanisms. For instance, sulfated polysaccharides derived from Porphyridium cruentum have demonstrated the ability to inhibit the replication of herpes simplex virus (HSV) and human immunodeficiency virus (HIV)24. The antiviral efficacy of these compounds is linked to their capacity to prevent viral entry into host cells, positioning them as promising candidates for antiviral therapy development. Recently, Pointcheval et al.25 examined the antimicrobial characteristics of exopolysaccharide-rich extracts from five microalgal species, which exhibited growth inhibition against both Gram- positive (Bacillus subtilis) and Gram-negative bacteria (Pseudomonas aeruginosa), as well as fungi (Cladosporium cladosporioides). The diverse bioactive properties of these extracts vary according to the specific microalgal species involved.
2.4 Phenolic Compounds
Microalgae, particularly Haematococcus pluvialis, are known to synthesize phenolic compounds, which encompass flavonoids, phenolic acids, and tannins. These compounds possess significant antioxidant and antimicrobial properties. They effectively neutralize reactive oxygen species (ROS) and impede the proliferation of pathogenic microorganisms by interfering with their cellular mechanisms. For instance, phenolic compounds derived from H. pluvialis have demonstrated the ability to inhibit the growth of Candida albicans, a prevalent fungal pathogen15.The combined antioxidant and antimicrobial functions of these compounds render them especially advantageous in the formulation of multifunctional therapeutic agents26. Research conducted by Alshuniaber et al.27, revealed that fraction B of the methanol extract from Spirulina is rich in polyphenols, which exhibit a broad spectrum of antimicrobial activity against drug-resistant foodborne bacterial pathogens. Additionally, various potential secondary metabolites, including benzophenone, dihydro-methyl-phenylacridine, carbanilic acid, dinitrobenzoate, propanediamine, isoquinoline, piperidine, oxazolidine, and pyrrolidine, have shown efficacy against both Gram-positive and Gram-negative pathogens.
2.5 Pigments
Microalgae serve as abundant reservoirs of pigments, including phycobiliproteins, chlorophylls, and carotenoids, which have been recognized for their antimicrobial properties28. For example, the green alga Chlorococcum humicola synthesizes pigments that exhibit efficacy against various bacterial pathogens, such as B. subtilis, S. aureus, and E. coli29. The antimicrobial effects of these pigments are believed to arise from their capacity to produce ROS within microbial cells, resulting in oxidative stress and subsequent cell death. Additionally, these pigments can compromise bacterial cell membranes by interacting with their lipid components, leading to cell leakage and ultimately cell lysis. Microalgae thus represent a significant and largely underexplored source of antimicrobial agents. Ongoing research and advancements in microalgal biotechnology will be essential for fully harnessing the potential of these extraordinary organisms.
3. Microalgae as a Source of Antibacterial Activity
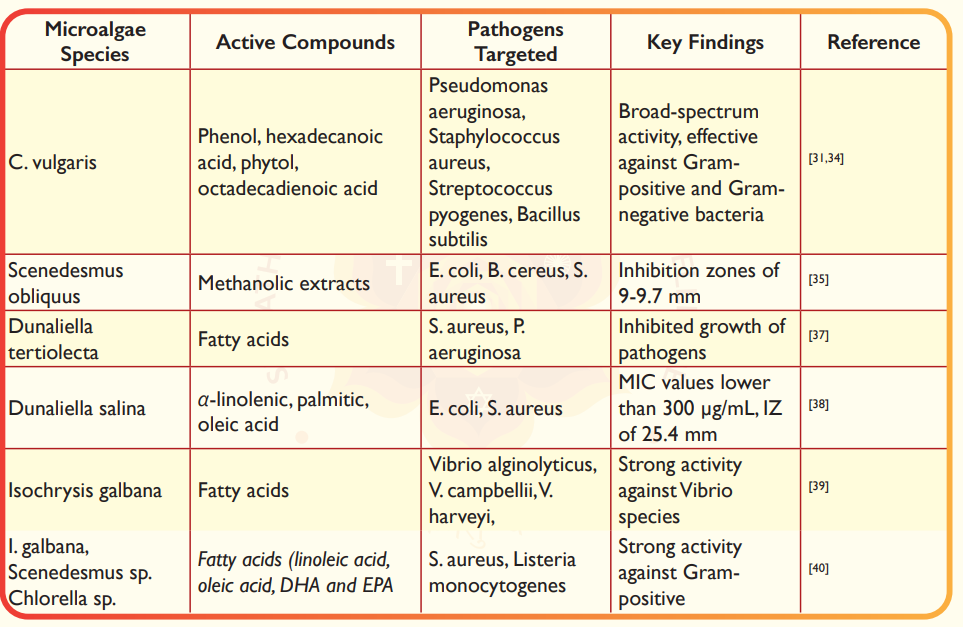
Microalgae have emerged as a promising source of bioactive compounds with antimicrobial properties, driven by the increasing burden of antibiotic resistance in humans30. These compounds have demonstrated significant potential in inhibiting a wide range of pathogenic bacteria, both Gram-positive and Gram-negative (Table 2). The first milestone in microalgal antibacterial research came with Chlorella vulgaris, from which bactericidal compounds were initially isolated, demonstrating effective inhibition against P. aeruginosa, S. aureus, Streptococcus pyogenes, and B. subtilis31. Further studies confirmed its broad-spectrum activity, with methanol extracts showing effectiveness against S. aureus, E. coli, B. subtilis, and B. cereus32,33. Notably, Chlorella sp. UKM8's methanol extract exhibited broad-spectrum activity against both Gram-positive and Gram-negative bacteria, attributed to compounds such as phenol (18.5%), hexadecanoic acid (18.25%), phytol (14.43%), and octadecadienoic acid (13.69%)34. Methanol extracts of Scenedesmus obliquus demonstrated activity against E. coli, B. cereus, and S. aureus, producing inhibition zones of 9-9.7 mm35. Additionally, whole-cell applications of Scenedesmus spp. cells eliminated Salmonella enterica growth within 48 hours, though the mechanism remained unclear36. Extracts from Dunaliella tertiolecta inhibited S. aureus and P. aeruginosa37, while D. salina extracts, attributed to fatty acids like α-linolenic, palmitic, and oleic acid, showed activity against E. coli, and S. aureus38. The microalga Isochrysis galbana synthesized antibacterial fatty acids that notably limited the growth of pathogenic Vibrio species such as V. alginolyticus, V. campbellii, and V. harveyi, except V. parahaemolyticus39.
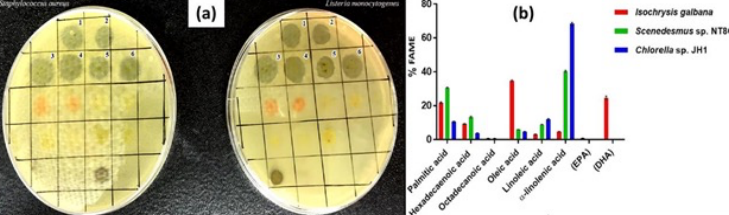
Alsenani et al.,40 found strong antibacterial activity against Gram-positive bacteria than Gram-negative bacteria from the microalgae extracts of Isochrysis galbana, Scenedesmus sp. and Chlorella sp., and identified and purified the fatty acids of linoleic acid, oleic acid, docosahexaenoicacid (DHA) and eicosapentaenoic acid (EPA) from the extract (Figure 3). Similarly, Phaeodactylum tricornutum extracts, particularly hexadecatrienoic acid (HTA), were active against S. aureus, including multidrug-resistant strains (MRSA)41. T. suecica extracts, containing fatty acids like methyl caprate and palmitic acid, elicited growth inhibition on Streptococcus pyogenes42. Short-chain fatty acids from H. pluvialis ethanolic extract also showed antimicrobial activity against Gram-negative E. coli43. C. vulgaris peptides inhibited P. aeruginosa growth15, while Dunaliella tertiolecta extracts also demonstrated activity against this pathogen37. Nannochloropsis oceanica, Isochrysis sp., and Thalassiosira weissflogii also showed promising results against V. harveyi44.
Ali and Doumandji45 found methanol extract from Spirulina exhibited widespread spectrum of antimicrobial activities against Gram-positive bacteria (43 ± 4.24 mm) and minimum inhibitory concentrations (MIC) 128 ± 0.71 µg/mL. Organic extracts from Chlorella excreted a broad spectrum of antimicrobial substances against Gram-negative bacteria. Recently, Ilieva et al.,46 discussed Arthrospira platensis with very potent antibacterial activity and minimum inhibitory concentrations (MICs) as low as 2-15 µg/mL against bacterial fish pathogens including Bacillus and Vibrio spp., also demonstrated an inhibition zone (IZ) of 50 mm against S. aureus. D. salina exhibited MIC values lower than 300 µg/mL and an IZ value of 25.4 mm on different bacteria, while D. tertiolecta showed MIC values of 25 and 50 µg/mL against some Staphylococcus spp. These values fulfill the criteria for significant antibacterial activity and sometimes are comparable or exceed the activity of the control antibiotics. Fusiform morphotypes of P. tricornutum expressed higher antibacterial activity than oval morphotypes, attributed to high levels of palmitoleic acid and other bioactive fatty acids22. Interestingly, polysaccharides from Chlamydomonas reinhardtii showed promising anti- biofilm potential by preventing biofilm formation and dissolving existing biofilms47.
Influence of methanol extraction has proven effective for isolating antimicrobial components from microalgae48. For example, Chlorella methanolic extracts showed antimicrobial activity against various bacteria with MIC values ranging from 2.6 to 5 mg/mL49. Short-chain fatty acids from H. pluvialis ethanolic extract showed antimicrobial activity against E. coli and S. aureus43. Mixed solvents (methanol:acetone:diethyl ether mixture) extracts of desert-sourced C. vulgaris and D. salina demonstrated broad-spectrum antibacterial activity50. Environmental conditions may influence antibacterial properties, as demonstrated by Cosmarium sp., from hot springs showing significant activity against various bacteria51, while Cosmarium laeve from non-extreme environments showed minimal activity. This indicates that stressed microalgae produce secondary metabolites with bioactive molecules, particularly with antimicrobial activities52.
Further research has shown the efficacy of microalgal compounds against drug-resistant ESKAPE germs. For example, methanol extracts from Chlorella vulgaris stopped the growth of drug-resistant Klebsiella pneumoniae. The lowest amount needed to inhibit growth was 3.2 mg/mL33. Also fatty acid (palmitoleic acid) extracts from Phaeodactylum tricornutum were effective against methicillin-resistant Staphylococcus aureus (MRSA) and Acinetobacter baumannii. The lowest amounts needed ranged from 50-100 µg/mL41. Furthermore, Scenedesmus obliquus extracts, which have phenolic compounds and fatty acids, showed good results (inhibition) against Pseudomonas aeruginosa35.These results show that microalgal compounds could be new treatments for ESKAPE germs, and warrant continued investigations on their mechanism of action and for clinical use.
Figure 3. Reproduced with permission from Alsenani et al., (2020)40, (a), Inhibition zones of Staphylococcus aureus and Listeria monocytogenes growth. 1: Isochrysis galbana crude extract; 2: I. galbana n-hexane fraction; 3: Scenedesmus sp. crude extract; 4: Scenedesmus sp. n-hexane fraction; 5: Chlorella sp. crude extract; 6: Chlorella sp. n-hexane fraction. (b), Percentage of individual fatty acid methyl esters (FAME) detected in each microalgal species.
3.1 Microalgal Compounds Against Biofilm Infections
Microalgal compounds have shown promise to fight biofilm infections, which resist antibiotics. Polysaccharides from C. reinhardtii have an impact on anti-biofilm activity. They stop biofilm formation and break up existing P. aeruginosa biofilms. At 100 µg/mL, they cut biofilm biomass by 60%47.Also methanolic extracts from Scenedesmus obliquus full of phenolic compounds, prevent S. aureus from forming biofilms. They reduce biofilm sticking by 70%35. These compounds likely alter or interfere with quorum sensing and the strength of the extracellular matrix, which are key to biofilm stability. This area also encourages continued research on mechanistic aspects and for their effective use in hospitals for long-lasting infections linked to medical devices.
Table 2. Antibacterial activity of microalgae, their active compounds, and the targeted bacterial pathogens
4. Microalgal Antifungal Activity
Fungal diseases are among the deadliest contagious diseases, causing approximately 1.5 million deaths annually53. Recent outbreaks, such as mucormycosis (black fungus), have highlighted the urgent need for effective antifungal treatments54. Despite the potential of microalgae as a source of antifungal agents, only a limited number of studies have explored this area, even though over 400 fungal species are known to act as opportunistic human pathogens55. The detrimental effects of fungal infections, such as black gill infections, allergic reactions, and asthmatic diseases, underscore the need for more comprehensive screening of microalgal species to identify potential antifungal compounds55.
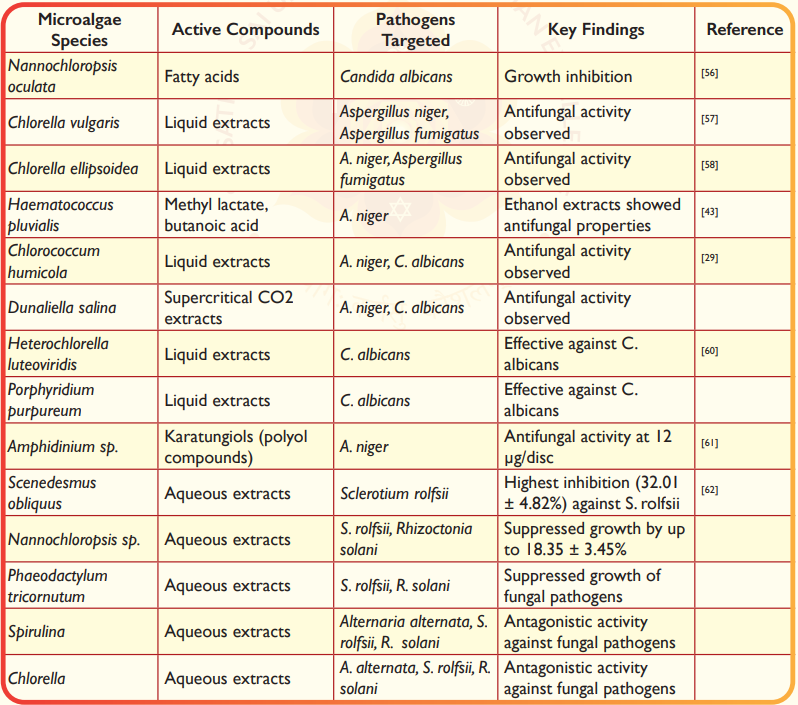
Microalgae have shown promising antifungal properties against a range of fungal species (Table 3). For instance, fatty acids derived from Nannochloropsis oculata have been shown to inhibit the growth of C. albicans56. Similarly,liquid extracts of C. vulgaris and Chlorella ellipsoidea exhibited antifungal activity against Aspergillus niger and Aspergillus fumigatus57,58. Ethanol extracts of H. pluvialis, containing methyl lactate and butanoic acid, also demonstrated antifungal properties against A. niger43. Several strains of microalgae isolated from freshwater lakes in Turkey have shown antifungal activity against Saccharomyces cerevisiae, C. albicans, Candida tropicalis, and Chlorococcus sp.59. Additionally, liquid extracts of Chlorococcum humicola and supercritical CO2 extracts from D. salina have shown antifungal activity against A. niger and C. albicans29. Furthermore, liquid extracts of Heterochlorella luteoviridis and Porphyridium purpureum have been effective against C. albicans60. Karatungiols, a novel antimicrobial polyol compounds, were isolated from the cultivated symbiotic marine dinoflagellate Amphidinium sp., exhibited antifungal activity against A. niger at 12 µg/disc61. Aqueous extracts from microalgal species such as Spirulina, Chlorella, Nannochloropsis, Scenedesmus, and P. tricornutum have shown antagonistic activity against fungal pathogens like Alternaria alternata, Sclerotium rolfsii, and Rhizoctonia solani in vitro. Among these, Scenedesmus obliquus exhibited the highest inhibition against S. rolfsii (32.01 ± 4.82%), while Nannochloropsis sp. and P. tricornutum suppressed the growth of S. rolfsii and R. solani by up to18.35 ± 3.45%62. These results suggest that microalgae could serve as sustainable alternatives to chemical fungicides in agriculture.
Table 3. Antifungal activity of microalgae, their active compounds, and the targeted fungal pathogens
5. Microalgal Antiviral Activity
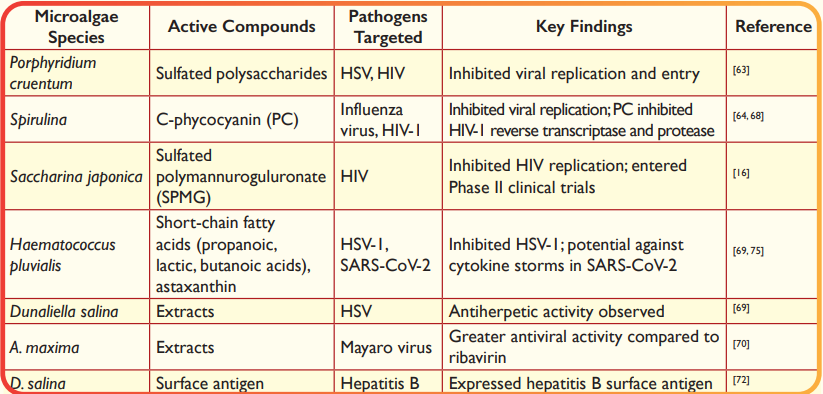
Microalgae-derived compounds have demonstrated significant antiviral activity against a range of viruses, including herpes simplex virus (HSV), human immunodeficiency virus (HIV), influenza virus, and SARS-CoV-2 (Table 4) and shown potential development of antiviral therapies and vaccines. Sulfated polysaccharides from Porphyridium cruentum have shown antiviral activity against HSV and HIV63. Similarly, Spirulina extracts have been reported to inhibit the replication of influenza virus64. China's first anti-AIDS drug, a heparin-like sulfated polysaccharide (sulfated polymannuroguluronate, SPMG) extracted from the brown macroalga Saccharina japonica, has entered Phase II clinical trials. This compound inhibits HIV replication and interferes with HIV entry into host T lymphocytes16. The inhibitory effects of microalgae-based bioactive metabolites are often due to their interaction with the positive charge on the virus's cell surface, preventing penetration into the host cell. Alternatively, these compounds may inhibit viral genome transcription or obstruct the formation of new virus particles65-67. Spirulina pigments C-phycocyanin (PC) has demonstrated unique antiviral properties against HIV-1 by inhibiting reverse transcriptase and protease enzymes. A concentration of 0.356 mg/mL of PC was found to inhibit HIV-1 replication by 80% while remaining safe for normal cells68. Ethanol extracts from H. pluvialis have demonstrated strong inhibition of herpes simplex virus type 1 (HSV-1) infection. The antiviral activity is attributed to short-chain fatty acids such as propanoic, lactic, and butanoic acids, as well as palmitic acid, hexadecatrienoic acid, and α-linolenic acid69. Microalgal species such as H. pluvialis and D. salina have also shown antiherpetic activity69. Extracts from A. maxima demonstrated greater antiviral activity compared to the commercial antiviral ribavirin. The study involved culturing four microalgae strains and testing their antiviral effects in vitro, revealing all strains had anti-Mayaro activity70. Compounds such as α- and β-ionone, neophytadiene, β-cyclocitral, and phytol extracted from microalgae have demonstrated antiviral properties71.These compounds further underscore the potential of microalgae as a source of bioactive metabolites for antiviral applications. Algae-derived vaccines are being explored for their potential in treating viral infections. For example, D. salina has been used to express a surface antigen for hepatitis B treatment72, and Chlamydomonas has been engineered to produce malaria vaccine antigens73. Spirulina- enriched diets have shown antiviral effects against HIV, improved insulin sensitivity, and regulated IL-6 and lipoprotein lipase activity. Immulina, a Spirulina extract, enhances immunological functions by activating toll-like receptors74. H. pluvialis, enriched with astaxanthin, has shown potential in reducing acute lung injury (ALI) and acute respiratory distress syndrome (ARDS). This suggests probable actions against cytokine storms caused by SARS-CoV-2 by increasing lymphocytes and reducing oxidative damage or decreasing IL-6 activity75.76.
Table 4. Antiviral activity of microalgae, their active compounds, and the viral targets
6. Microalgal Antiprotozoan Activity
Microalgae have demonstrated significant antiprotozoan activity against neglected tropical diseases (NTDs) such as leishmaniasis, Chagas disease, and human African trypanosomiasis (HAT). These diseases are caused by protozoan parasites, including Leishmania spp., Trypanosoma cruzi, and Trypanosoma brucei77. The bioactive compounds derived from microalgae show promise as potential treatments for these diseases (Table 5), addressing the urgent need for effective and safe therapies.
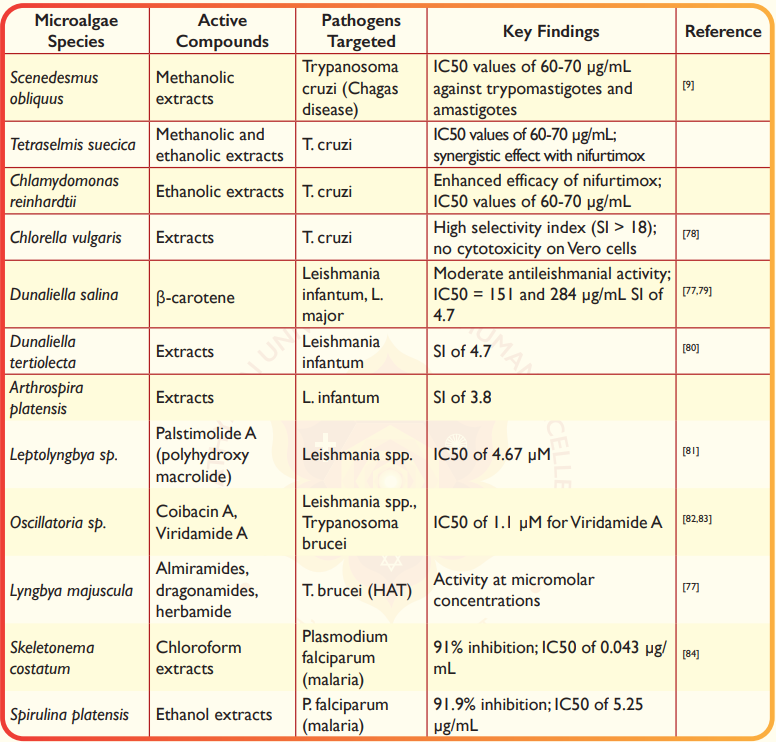
Microalgal extracts have shown significant trypanocidal activity against T. cruzi, the causative agent of Chagas disease. Methanol extracts of S. obliquus and T. suecica, as well as ethanol extracts of C. reinhardtii and T. suecica, demonstrated trypanocidal activity against both extracellular trypomastigotes and intracellular amastigotes, with IC₅₀ values ranging from 60 to 70 µg/mL9. The ethanol extract of C. reinhardtii was found to enhance the efficacy of the conventional antichagasic drug nifurtimox, suggesting a potential synergistic effect9. Additionally, C. vulgaris and Tetradesmus obliquus have shown significant trypanocidal activity against T. cruzi. C. vulgaris, in particular, demonstrated a high selectivity index (SI > 18) and no cytotoxic effects on Vero cells, making it a promising candidate for drug development78. Microalgae have also demonstrated antileishmanial activity against Leishmania spp., the causative agents of leishmaniasis. For instance, D. salina showed moderate antileishmanial activity, which may be attributed to its high β-carotene content77. Gharbi et al.79 identified Dunaliella sp. from Tunisian water bodies, highlighting its promising antileishmanial activity against L. infantum and L. major (IC₅₀=151 and 284 µg/mL, respectively). Similarly, D. tertiolecta and A. platensis extracts demonstrated a selectivity index (SI) of 4.7 and 3.8 against L. infantum, outperforming meglumine antimoniate (SI=2.1), respectively80. Cyanobacteria have also been a source of antileishmanial compounds. Palstimolide A, a complex polyhydroxy macrolide isolated from Leptolyngbya sp., showed significant antileishmanial activity with an IC₅₀ of 4.67 µM81. Coibacin A, derived from Oscillatoria sp., also demonstrated antileishmanial properties82. Viridamide A, isolated from Oscillatoria nigro-viridis, showed antitrypanosomal activity with an IC₅₀ of 1.1 µM83. Additionally, peptides such as almiramides, dragonamides, and herbamide, biosynthesized by Lyngbya majuscula, exhibited activity against T. brucei at micromolar concentrations77. Microalgal extracts have also shown antiplasmodial activity against Plasmodium falciparum, the causative agent of malaria. The chloroform extract of Skeletonema costatum demonstrated the highest inhibitory activity (91% inhibition) with an IC₅₀ of 0.043 µg/mL, while the ethanol extract of S. platensis showed 91.9% inhibition with an IC₅₀ of 5.25 µg/mL84. Despite the promising results, no natural microalgae products or their derivatives have entered clinical testing for antiprotozoan activity. Further research is needed to isolate and characterize bioactive compounds from microalgae and cyanobacteria, evaluate their safety and efficacy, and explore their potential as novel treatments for NTDs.
Table 5. Antiprotozoan activity of microalgae, their active compounds, and the protozoan parasite targets
7. Challenges and Future Directions
Microalgae show real promise as antimicrobial powerhouses, yet there are hurdles before their potential is fully harnessed. There is a need to fine-tune their growth conditions to maximize the yield of bioactive compound production, develop better extraction and purification methods, and establish rigor in testing their safety and efficacy. Another challenge is that many microalgae derived compounds have complex structures that make large-scale production tricky. The good news is that breakthroughs in genetic engineering and synthetic biology could be game-changers. Imagine tweaking microalgae DNA to pump out more antimicrobial compounds or even engineer entirely new ones, which could dramatically boost both yields and variety. Pairing microalgae farming with bio-refinery techniques might also offer a cost-effective, eco-friendly way to scale up production. Interestingly, research into antifungal resistance has trailed behind antibacterial studies, partly because fungal infections weren't seen as major threats until recently. But the stakes are high for example, deaths from candidiasis have surged due to modern medical practices like use of immunosuppressive therapies and broadspectrum antibiotics85. Ability of microalgae to produce diverse bioactive molecules makes them exciting candidates for next-gen antifungals and other antimicrobials. However, there is imminent need for extensive research to unlock their full potential both for fighting human infections and protecting crops; and to develop sustainable solutions that actually work in the real world.
7.1 Bioprocesses and Bioreactor Design for Antimicrobial Production
Producing antimicrobial compounds from microalgae needs custom bioprocesses and bioreactor designs to boost yield and cut costs. Photobioreactors built to improve light penetration and CO2 delivery, play a key role in growing microalgae. These systems must keep ideal conditions like light intensity (100-200 µmol photons m⁻² s⁻¹), pH (7-9), and temperature (20-30°C) to boost biomass and metabolite production14. Unlike bacterial fermenters, photobioreactors don't need organic carbon sources, which might lower running costs. But, issues like high energy costs for mixing and lighting, plus the need for good harvesting and extraction methods, increase financial implications. Current studies focus on using bio-refinery approaches where they extract many products (e.g., biofuels, pigments, and antimicrobials) from microalgal biomass to improve cost-effectiveness11. To scale up production, better bioreactor designs and process improvements to make microalgal antimicrobials compete with synthetic options emerge as the need of the hour.
7.2 Safety and Efficacy of Microalgal Compounds
The potential of microalgae-derived antimicrobial compounds to treat diseases faces challenges due to the lack of thorough safety and efficacy studies. Lab tests show these compounds can fight various germs, but research on living organisms or in clinical settings is limited. Sulfated polysaccharides from Porphyridium cruentum which can combat HSV and HIV viruses without harming cells24 is a good example. However, not enough is known about how they move through and might affect the human body. In the same way, peptides from Chlorella vulgaris can kill bacteria, but if they trigger immune responses or cause harm throughout the body needs to be established20. Rules set by agencies like the Food and Drug Administration (FDA) require a lot of testing prior to and during clinical trials to ensure these compounds are safe. This includes looking at short-term and long-term toxic effects, allergic reactions, and unintended impacts. Moving forward, researchers should focus on running standard toxicity tests and clinical trials to establish how safe these compounds are, which will help bring them into regular medical use.
7.3 Genetic Engineering for Enhanced Antimicrobial Production
Researchers explore new ways to use genetic engineering and synthetic biology to get microalgae to produce more and different types of antimicrobial compounds. They apply novel genome editing tools like CRISPRCas9, RNAi, ZNFs, TALENs and synthetic biology to adjust microalgal genomes. This allows them to improve processes inside the cells that create useful substances. Researchers enhanced genes linked to enzymes, lipid synthesis and pigment production86,87. Nuclear engineering allows protein secretion and post-translational modifications, such as glycosylation, while chloroplast engineering ensures high-level, stable expression without these modifications88. Chlamydomonas reinhardtii has emerged as a key model, producing therapeutic proteins like endolysins at ~1% of total soluble protein. Diatoms, such as Thalassiosira pseudonana, have been engineered to produce vaccines, with yields enhanced by conditions like silicon limitation. Techniques like codon optimization and synthetic promoters have boosted expression levels, and companies like Triton Health and Nutrition are advancing commercial production88. While these advancements are promising, challenges remain. Complicated microalgal genomes and fears of unintended genetic effects add to the difficulty. GMO regulations also create obstacles. Using techniques such as high-throughput screening and metabolic modeling may help make engineered microalgae more practical to produce antimicrobial compounds on a large scale.
7.4 Potential for Resistance to Microalgal Compounds
The rise of resistance to antimicrobial compounds from microalgae also poses a serious problem, but research is still limited as the field is new. Conventional antibiotics target one specific pathway, while microalgal compounds like antimicrobial peptides (AMPs) and fatty acids work in several ways. They disrupt membranes and cause oxidative stress, which might make resistance harder to develop18. AMPs from Nostoc species, for instance, break down bacterial membranes in a broad non-specific way making it tough for bacteria to build resistance19. Still long-term exposure might push bacteria to adapt, like by activating efflux pumps or changing their membrane structure. The need to study resistance patterns, test combination treatments, and perform detailed investigations to reduce this risk and keep microalgal compounds effective for the future remains high.
8. Conclusion
Microalgae constitute a large, mostly underutilized source of bioactive substances with considerable antimicrobial capabilities. Their capacity to generate a varied range of metabolites such as peptides, fatty acids, polysaccharides, phenolic compounds, and pigments makes them a valuable alternative in combating antibiotic-resistant pathogens. Nevertheless, issues like enhancing their cultivation conditions, refining extraction techniques, and guaranteeing the safety and efficacy of these compounds need to be tackled to completely leverage their therapeutic capabilities. Developments in genetic engineering and synthetic biology present exciting prospects for improving the production and variety of these bioactive metabolites. Intense research especially in vivo studies with interdisciplinary collaboration will be crucial to realize the complete potential of microalgae, leading to sustainable and effective antimicrobial treatments in the future.
Author Contributions: Conceptualization - A.S; N.K.S and S.K.S; Methodology - A.S; writing- original draft preparation - A.S; N.K.S and S.K.S; writing-review and editing - A.S; N.K.S and S.K.S.
Funding: This research received no external funding
Institutional Review Board Statement: Not applicable for studies not involving humans or animals.
Data Availability Statement: No new data were created
Acknowledgments: The authors are grateful to the University for enabling us to pursue microalgae research in the Life Science department.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Suresh, A.; Benor, S. Microalgae-based biomass production for control of air pollutants. In: Biofiltration to Promising Options in Gaseous Fluxes Biotreatment; Elsevier: 2020; pp. 345–372. https://doi.org/10.1016/B978-0-12-819064-7.00017-0
2. Dimopoulou, M.; Kolonas, A.; Stagos, D.; Gortzi, O. A Review of the Sustainability, Chemical Composition, Bioactive Compounds, Antioxidant and Antidiabetic Activity, Neuroprotective Properties, and Health Benefits of Microalgae. Biomass 2025, 5(1), 11. https://doi.org/10.3390/biomass5010011
3. Dhandwal, A.; Bashir, O.; Malik, T.; Salve, R.V.; Dash, K.K.; Amin, T.; Shams, R.; Wani, A.W.; Shah, Y.A. Sustainable microalgal biomass as a potential functional food and its applications in food industry: A comprehensive review. Environ. Sci. Pollut. Res. 2024, 31(1), 1–19. https://doi.org/10.1007/s11356-024-33431-6
4. Singh, R.; Parihar, P.; Singh, M.; Bajguz, A.; Kumar, J.; Singh, S.; Singh, V.P.; Prasad, S.M. Uncovering potential applications of cyanobacteria and algal metabolites in biology, agriculture and medicine: Current status and future prospects. Front. Microbiol. 2017, 8, 515. https://doi.org/10.3389/fmicb.2017.00515
5. Bratchkova, A.; Kroumov, A.D. Microalgae as producers of biologically active compounds with antibacterial, antiviral, antifungal, antialgal, antiprotozoal, antiparasitic and anticancer activity. Acta Microbiol. Bulg. 2020, 36(3), 79–89.
6. Stirk, W.A.; van Staden, J. Bioprospecting for bioactive compounds in microalgae: Antimicrobial compounds. Biotechnol. Adv. 2022, 59, 107977. https://doi.org/10.1016/j.biotechadv.2022.107977
7. Yarkent, Ç., Aslanbay Güler, B., Imamoglu, E. and Oncel, S.S., 2024. Microalgae-factories as potential antimicrobial agents: a comprehensive review. Biologia, 79(5), pp.1237-1250. https://doi.org/10.1007/s11756-024-01616-6
8. Najdenski, H.M.; Gigova, L.G.; Iliev, I.I.; Pilarski, P.S.; Lukavský, J.; Tsvetkova, I.V. Antibacterial and antifungal activities of selected microalgae and cyanobacteria. Int. J. Food Sci. Technol. 2013, 48(8), 1533–1540. https://doi.org/10.1111/ijfs.12122
9. Veas, R.; Rojas-Pirela, M.; Castillo, C.; Olea-Azar, C.; Moncada, M.; Ulloa, P.; Rojas, V.; Kemmerling, U. Microalgae extracts: Potential anti-Trypanosoma cruzi agents? Biomed. Pharmacother. 2020, 127, 110178. https://doi.org/10.1016/j.biopha.2020.110178
10. Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101(2), 87–96. https://doi.org/10.1263/jbb.101.87
11. Caporgno, M.P.; Mathys, A. Trends in microalgae incorporation into innovative food products with potential health benefits. Front. Nutr. 2018, 5, 58. https://doi.org/10.3389/fnut.2018.00058
12. Sathasivam, R.; Radhakrishnan, R.; Hashem, A.; Abd Allah, E.F. Microalgae metabolites: A rich source for food and medicine. Saudi J. Biol. Sci. 2019, 26(7), 709–722. https://doi.org/10.1016/j.sjbs.2017.11.003
13. Little, S.M.; Senhorinho, G.N.; Saleh, M.; Basiliko, N.; Scott, J.A. Antibacterial compounds in green microalgae from extreme environments: A review. Algae 2021, 36(1), 61–72. https://doi.org/10.4490/algae.2021.36.3.6
14. Borowitzka, M.A. High-value products from microalgae—Their development and commercialisation. J. Appl. Phycol. 2013, 25(3), 743–756. https://doi.org/10.1007/s10811-013-9983-9
15. Guedes, A.C.; Amaro, H.M.; Malcata, F.X. Microalgae as sources of high added‐value compounds—A brief review of recent work. Biotechnol. Prog. 2011, 27(3), 597–613. https://doi.org/10.1002/btpr.575
16. Dai, N.; Wang, Q.; Xu, B.; Chen, H. Remarkable natural biological resource of algae for medical applications. Front. Mar. Sci. 2022, 9, 912924. https://doi.org/10.3389/fmars.2022.912924
17. Nunes, E.; Odenthal, K.; Nunes, N.; Fernandes, T.; Fernandes, I.A.; de Carvalho, M.A.P. Protein extracts from microalgae and cyanobacteria biomass. Techno-functional properties and bioactivity: A review. Algal Res. 2024, 103638. https://doi.org/10.1016/j.algal.2024.103638
18. Ayswaria, R.; Vijayan, J.; Nathan, V.K. Antimicrobial peptides derived from microalgae for combating antibiotic resistance: Current status and prospects. Cell Biochem. Funct. 2023, 41(2), 142–151. https://doi.org/10.1002/cbf.3779
19. Dembitsky, V.M.; Řezanka, T. Metabolites produced by nitrogen-fixing Nostoc species. Folia Microbiol. 2005, 50(4), 363–391. https://doi.org/10.1007/BF02931419
20. Guzmán, F.; Wong, G.; Román, T.; Cárdenas, C.; Alvárez, C.; Schmitt, P.; Albericio, F.; Rojas, V. Identification of antimicrobial peptides from the microalgae Tetraselmis suecica (Kylin) Butcher and bactericidal activity improvement. Mar. Drugs 2019, 17(8), 453. https://doi.org/10.3390/md17080453
21. Desbois, A.P.; Smith, V.J. Antibacterial free fatty acids: Activities, mechanisms of action and biotechnological potential. Appl. Microbiol. Biotechnol. 2010, 85(6), 1629–1642. https://doi.org/10.1007/s00253-009-2355-3
22. Desbois, A.P.; Walton, M.; Smith, V.J. Differential antibacterial activities of fusiform and oval morphotypes of Phaeodactylum tricornutum (Bacillariophyceae). J. Mar. Biol. Assoc. U.K. 2010, 90(4), 769–774. https://doi.org/10.1017/S0025315409991366
23. Navarro, F.; et al. Antimicrobial activity of the acidophilic eukaryotic microalga Coccomyxa onubensis. J. Phycol. Res. 2017, 65(1), 38–43. https://doi.org/10.1111/pre.12158
24. Raposo, M.F.J.; de Morais, R.M.S.C.; de Morais, A.M.M.B. Bioactivity and applications of sulphated polysaccharides from marine microalgae. Mar. Drugs 2013, 11(1), 233–252. https://doi.org/10.3390/md11010233
25. Pointcheval, M.; Massé, A.; Floc’hlay, D.; Chanonat, F.; Estival, J.; Durand, M.J. Antimicrobial properties of selected microalgae exopolysaccharide-enriched extracts: influence of antimicrobial assays and targeted microorganisms. Front. Microbiol. 2025, 16, 1536185. https://doi.org/10.3389/fmicb.2025.1536185
26. Cichoński, J.; Chrzanowski, G. Microalgae as a source of valuable phenolic compounds and carotenoids. Molecules 2022, 27(24), 8852. https://doi.org/10.3390/molecules27248852
27. Alshuniaber, M.A.; Krishnamoorthy, R.; AlQhtani, W.H. Antimicrobial activity of polyphenolic compounds from Spirulina against food-borne bacterial pathogens. Saudi J. Biol. Sci. 2021, 28(1), 459–464. https://doi.org/10.1016/j.sjbs.2020.10.029
28. Papadaki, S.; Tricha, N.; Panagiotopoulou, M.; Krokida, M. Innovative Bioactive Products with Medicinal Value from Microalgae and Their Overall Process Optimization through the Implementation of Life Cycle Analysis—An Overview. Mar. Drugs 2024, 22(4), 152. https://doi.org/10.3390/md22040152
29. Bhagavathy, S.; Sumathi, P.; Bell, I.J.S. Green algae Chlorococcum humicola—A new source of bioactive compounds with antimicrobial activity. Asian Pac. J. Trop. Biomed. 2011, 1(1), S1–S7. https://doi.org/10.1016/S2221-1691(11)60111-1
30. Shannon, E.; Abu-Ghannam, N. Antibacterial derivatives of marine algae: An overview of pharmacological mechanisms and applications. Mar. Drugs 2016, 14(4), 81. https://doi.org/10.3390/md14040081
31. Pratt, R.; Daniels, T.C.; Eiler, J.J.; Gunnison, J.B.; Kumler, W.D.; Oneto, J.F.; Strait, L.A.; Spoehr, H.A.; Hardin, G.J.; Milner, H.W.; Smith, J.H.C. Chlorellin, an antibacterial substance from Chlorella. Science 1944, 99(2574), 351–352. https://doi.org/10.1126/science.99.2574.351
32. Kellam, S.J.; Walker, J.M. Antibacterial activity from marine microalgae in laboratory culture. Br. Phycol. J. 1989, 24(2), 191–194. https://doi.org/10.1080/00071618900650181
33. Salem, W.M.; Galal, H.; Nasr El-deen, F. Screening for antibacterial activities in some marine algae from the red sea (Hurghada, Egypt). Afr. J. Microbiol. Res. 2011, 5(15), 2160–2167.
34. Shaima, A.F.; Yasin, N.H.M.; Ibrahim, N.; Takriff, M.S.; Gunasekaran, D.; Ismaeel, M.Y. Unveiling antimicrobial activity of microalgae Chlorella sorokiniana (UKM2), Chlorella sp. (UKM8) and Scenedesmus sp. (UKM9). Saudi J. Biol. Sci. 2022, 29(2), 1043–1052. https://doi.org/10.1016/j.sjbs.2021.09.069
35. Marrez, D.A.; Naguib, M.M.; Sultan, Y.Y.; Higazy, A.M. Antimicrobial and anticancer activities of Scenedesmus obliquus metabolites. Heliyon 2019, 5(3). https://doi.org/10.1016/j.heliyon.2019.e01404
36. Mezzari, M. P., J. M. Prandini, J. Deon Kich, and M. L. B. D. Silva. "Elimination of antibiotic multi-resistant Salmonella typhimurium from swine wastewater by microalgae-induced antibacterial mechanisms." J. Bioremediat. Biodegrad 8 (2017): 379. http://dx.doi.org/10.4172/2155-6199.1000379
37. Pane, G.; Cacciola, G.; Giacco, E.; Mariottini, G.L.; Coppo, E. Assessment of the antimicrobial activity of algae extracts on bacteria responsible of external otitis. Mar. Drugs 2015, 13(10), 6440–6452. https://doi.org/10.3390/md13106440
38. Herrero, M.; Jaime, L.; Martín-Álvarez, P.J.; Cifuentes, A.; Ibáñez, E. Optimization of the extraction of antioxidants from Dunaliella salina microalga by pressurized liquids. J. Agric. Food Chem. 2006, 54(15), 5597–5603. https://doi.org/10.1021/jf060546q
39. Molina-Cárdenas, C.A.; Sánchez-Saavedra, M.D.P.; Lizárraga-Partida, M.L. Inhibition of pathogenic Vibrio by the microalgae Isochrysis galbana. J. Appl. Phycol. 2014, 26(6), 2347–2355. https://doi.org/10.1007/s10811-014-0270-1
40. Alsenani, F.; Tupally, K.R.; Chua, E.T.; Eltanahy, E.; Alsufyani, H.; Parekh, H.S.; Schenk, P.M. 2020. Evaluation of microalgae and cyanobacteria as potential sources of antimicrobial compounds. Saudi Pharmaceutical Journal, 2020, 28(12), pp.1834-1841. https://doi.org/10.1016/j.jsps.2020.11.010
41. Desbois, A.P.; Lebl, T.; Yan, L.; Smith, V.J. Isolation and structural characterisation of two antibacterial free fatty acids from the marine diatom, Phaeodactylum tricornutum. Appl. Microbiol. Biotechnol. 2008, 81(4), 755–764. https://doi.org/10.1007/s00253-008-1714-9
42. Bai, V.D.M.; Krishnakumar, S. Evaluation of antimicrobial metabolites from marine microalgae Tetraselmis suecica using gas chromatography-mass spectrometry (GC-MS) analysis. Int. J. Pharm. Pharm. Sci. 2013, 5(3), 17–23.
43. Santoyo, S.; Rodríguez-Meizoso, I.; Cifuentes, A.; Jaime, L.; Reina, G.G.B.; Señorans, F.J.; Ibáñez, E. Green processes based on the extraction with pressurized fluids to obtain potent antimicrobials from Haematococcus pluvialis microalgae. LWT-Food Sci. Technol. 2009, 42(7), 1213–1218. https://doi.org/10.1016/j.lwt.2009.01.012
44. Jusidin, M.R.; Othman, R.; Shaleh, S.R.M.; Ching, F.F.; Senoo, S.; Oslan, S.N.H. In vitro antibacterial activity of marine microalgae extract against Vibrio harveyi. Appl. Sci. 2022, 12(3), 1148. https://doi.org/10.3390/app12031148
45. Ali, I.; Doumandji, A. Comparative phytochemical analysis and in vitro antimicrobial activities of the cyanobacterium Spirulina platensis and the green alga Chlorella pyrenoidosa: potential application of bioactive components as an alternative to infectious diseases. Bull. Inst. Sci., Rabat, Sect. Sci. Vie 2017, 39, 41–49.
46. Ilieva, Y.; Zaharieva, M.M.; Najdenski, H.; Kroumov, A.D. Antimicrobial activity of Arthrospira (former Spirulina) and Dunaliella related to recognized antimicrobial bioactive compounds. Int. J. Mol. Sci. 2024, 25(10), 5548. https://doi.org/10.3390/ijms25105548
61. Washida, K.; Koyama, T.; Yamada, K.; Kita, M.; Uemura, D. Karatungiols A and B, two novel antimicrobial polyol compounds, from the symbiotic marine dinoflagellate Amphidinium sp. Tetrahedron Lett. 2006, 47(15), 2521–2525. https://doi.org/10.1016/j.tetlet.2006.02.045
62. Schmid, B.; Coelho, L.; Schulze, P.S.; Pereira, H.; Santos, T.; Maia, I.B.; Reis, M.; Varela, J. Antifungal properties of aqueous microalgal extracts. Bioresour. Technol. Rep. 2022, 18, 101096. https://doi.org/10.1016/j.biteb.2022.101096
63. de Jesus Raposo, M.F.; de Morais, A.M.M.B.; de Morais, R.M.S.C. Influence of sulphate on the composition and antibacterial and antiviral properties of the exopolysaccharide from Porphyridium cruentum. Life Sci. 2014, 101(1–2), 56–63. https://doi.org/10.1016/j.lfs.2014.02.013
64. Hayashi, T.; Hayashi, K.; Maeda, M.; Kojima, I. Calcium spirulan, an inhibitor of enveloped virus replication, from a blue-green alga Spirulina platensis. J. Nat. Prod. 1996, 59(1), 83–87. https://doi.org/10.1021/np960017o
65. Pradhan, B.; Nayak, R.; Patra, S.; Bhuyan, P.P.; Dash, S.R.; Ki, J.S.; Adhikary, S.P.; Ragusa, A.; Jena, M. Cyanobacteria and algae-derived bioactive metabolites as antiviral agents: Evidence, mode of action, and scope for further expansion; a comprehensive review in light of the SARS-CoV-2 outbreak. Antioxidants 2022, 11(2), 354. https://doi.org/10.3390/antiox11020354
66. Reis, J.G.; Cadamuro, R.D.; Cabral, A.C.; Thaís da Silva, I.; Rodríguez-Lázaro, D.; Fongaro, G. Broad spectrum algae compounds against viruses. Front. Microbiol. 2022, 12, 809296. https://doi.org/10.3389/fmicb.2021.809296
67. Reynolds, D.; Huesemann, M.; Edmundson, S.; Sims, A.; Hurst, B.; Cady, S.; Beirne, N.; Freeman, J.; Berger, A.; Gao, S. Viral inhibitors derived from macroalgae, microalgae, and cyanobacteria: A review of antiviral potential throughout pathogenesis. Algal Res. 2021, 57, 102331. https://doi.org/10.1016/j.algal.2021.102331
68. Jadaun, P.; Seniya, C.; Pal, S.K.; Kumar, S.; Kumar, P.; Nema, V.; Kulkarni, S.S.; Mukherjee, A. Elucidation of antiviral and antioxidant potential of C-phycocyanin against HIV-1 infection through in silico and in vitro approaches. Antioxidants 2022, 11(10), 1942. https://doi.org/10.3390/antiox11101942
69. Santoyo, S.; Jaime, L.; Plaza, M.; Herrero, M.; Rodriguez-Meizoso, I.; Ibañez, E.; Reglero, G. Antiviral compounds obtained from microalgae commonly used as carotenoid sources. J. Appl. Phycol. 2012, 24(3), 731–741. https://doi.org/10.1007/s10811-011-9692-1
70. Ribeiro, M.C.M.; Salles, T.S.; Moreira, M.F.; Barbarino, E.; do Valle, A.F.; Couto, M.A.P.G. Antiviral activity of microalgae extracts against Mayaro virus. Algal Res. 2022, 61, 102577. https://doi.org/10.1016/j.algal.2021.102577
71. Amaro, H.M.; Guedes, A.C.; Malcata, F.X. Antimicrobial activities of microalgae: An invited review. Sci. Against Microb. Pathog. 2011, 2, 1272–1284.
72. Geng, D.; Wang, Y.; Wang, P.; Li, W.; Sun, Y. Stable expression of hepatitis B surface antigen gene in Dunaliella salina (Chlorophyta). J. Appl. Phycol. 2003, 15(5), 451–456. https://doi.org/10.1023/B:JAPH.0000004298.89183.e5
73. Dauvillée, D.; Delhaye, S.; Gruyer, S.; Slomianny, C.; Moretz, S.E.; d’Hulst, C.; Long, C.A.; Ball, S.G.; Tomavo, S. Engineering the chloroplast targeted malarial vaccine antigens in Chlamydomonas starch granules. PLoS One 2010, 5(12), e15424. https://doi.org/10.1371/journal.pone.0015424
74. Kefayat, A.; Ghahremani, F.; Safavi, A.; Hajiaghababa, A.; Moshtaghian, J. Spirulina extract enriched for Braun-type lipoprotein (Immulina®) for inhibition of 4T1 breast tumors growth and metastasis. Phytother. Res. 2020, 34(2), 368–378. https://doi.org/10.1002/ptr.6527
75. Talukdar, J.; Dasgupta, S.; Nagle, V.; Bhadra, B. COVID-19: Potential of microalgae derived natural astaxanthin as adjunctive supplement in alleviating cytokine storm. SSRN 2020, 3579738. https://dx.doi.org/10.2139/ssrn.3579738
76. Carbone, D.A.; Pellone, P.; Lubritto, C.; Ciniglia, C. Evaluation of microalgae antiviral activity and their bioactive compounds. Antibiotics 2021, 10(6), 746. https://doi.org/10.3390/antibiotics10060746
77. Matos, Â.P.; Saldanha-Corrêa, F.M.P.; da Silva Gomes, R.; Hurtado, G.R. Exploring microalgal and cyanobacterial metabolites with antiprotozoal activity against Leishmania and Trypanosoma parasites. Acta Trop. 2024, 251, 107116. https://doi.org/10.1016/j.actatropica.2023.107116
78. Júnior, J.N.D.S.; da Silva, A.C.; Oliveira, K.K.D.S.; Moreira, L.R.; Caires, S.D.F.F.D.S.; da Silva, A.J.; Moura, Y.A.S.; Marques, D.D.A.V.; Bezerra, R.P.; de Lorena, V.M.B.; Porto, A.L.F. Green microalgae as a potential source of trypanocide compounds. Nat. Prod. Res. 2024, 38(13), 2329–2335. https://doi.org/10.1080/14786419.2023.2169688
79. Gharbi, K.; Fathalli, A.; Essid, R.; Fassatoui, C.; Romdhane, M.S.; Limam, F.; Jenhani, A.B.R. Tunisian inland water microflora as a source of phycobiliproteins and biological activity with beneficial effects on human health. Oceanol. Hydrobiol. Stud. 2021, 50(4), 385–397. https://doi.org/10.2478/oandhs-2021-0033
80. Vaitkevicius-Antão, V.; Moreira-Silva, J.; Reino, I.B.D.S.M.; Melo, M.G.N.D.; Silva-Júnior, J.N.D.; Andrade, A.F.D.; Araújo, P.S.R.D.; Bezerra, R.P.; Marques, D.D.A.V.; Ferreira, S.; Pessoa-e-Silva, R. Therapeutic potential of photosynthetic microorganisms for visceral leishmaniasis: An immunological analysis. Front. Immunol. 2022, 13, 891495. https://doi.org/10.3389/fimmu.2022.891495
81. Keller, L.; Siqueira-Neto, J.L.; Souza, J.M.; Eribez, K.; LaMonte, G.M.; Smith, J.E.; Gerwick, W.H. Palstimolide A: A complex polyhydroxy macrolide with antiparasitic activity. Molecules 2020, 25(7), 1604. https://doi.org/10.3390/molecules25071604
82. Carneiro, V.M.; Avila, C.M.; Balunas, M.J.; Gerwick, W.H.; Pilli, R.A. Coibacins A and B: Total synthesis and stereochemical revision. J. Org. Chem. 2014, 79(2), 630–642. https://doi.org/10.1021/jo402339y
83. Simmons, T.L.; Engene, N.; Ureña, L.D.; Romero, L.I.; Ortega-Barría, E.; Gerwick, L.; Gerwick, W.H. Viridamides A and B, lipodepsipeptides with antiprotozoal activity from the marine cyanobacterium Oscillatoria nigroviridis. J. Nat. Prod. 2008, 71(9), 1544–1550. https://doi.org/10.1021/np800110e
84. Setyowati, E.A.; Isnansetyo, A.; Djohan, T.S.; Nurcahyo, R.W. Antimalarial activity of microalgae extracts based on inhibition of PfMQO, a mitochondrial Plasmodium falciparum enzyme. Pharmacogn. J. 2019, 11(6s). http://dx.doi.org/10.5530/pj.2019.11.228
85. Garvey, M.; Meade, E.; Rowan, N.J. Effectiveness of front line and emerging fungal disease prevention and control interventions and opportunities to address appropriate eco-sustainable solutions. Sci. Total Environ. 2022, 851, 158284. https://doi.org/10.1016/j.scitotenv.2022.158284
86. Fayyaz, M.; Chew, K.W.; Show, P.L.; Ling, T.C.; Ng, I.S.; Chang, J.S. Genetic engineering of microalgae for enhanced biorefinery capabilities. Biotech. Adv. 2020, 43, 107554. https://doi.org/10.1016/j.biotechadv.2020.107554
87. Naduthodi, M.I.S.; Claassens, N.J.; D’Adamo, S.; van der Oost, J.; Barbosa, M.J. Synthetic biology approaches to enhance microalgal productivity. Trends. Biotech. 2021, 39(10), 1019-1036. https://doi.org/10.1016/j.tibtech.2020.12.010
88. Grama, S.B.; Liu, Z.; Li, J. Emerging trends in genetic engineering of microalgae for commercial applications. Mar. Drugs 2022, 20, 285. https://doi.org/10.3390/md20050285
Disclaimer/Publisher's Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of SSSUHE and/or the editor(s). SSSUHE and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions, or products referred to in the content.